Ferric Nitrate Formula: Properties, Uses, and Applications
Introduction
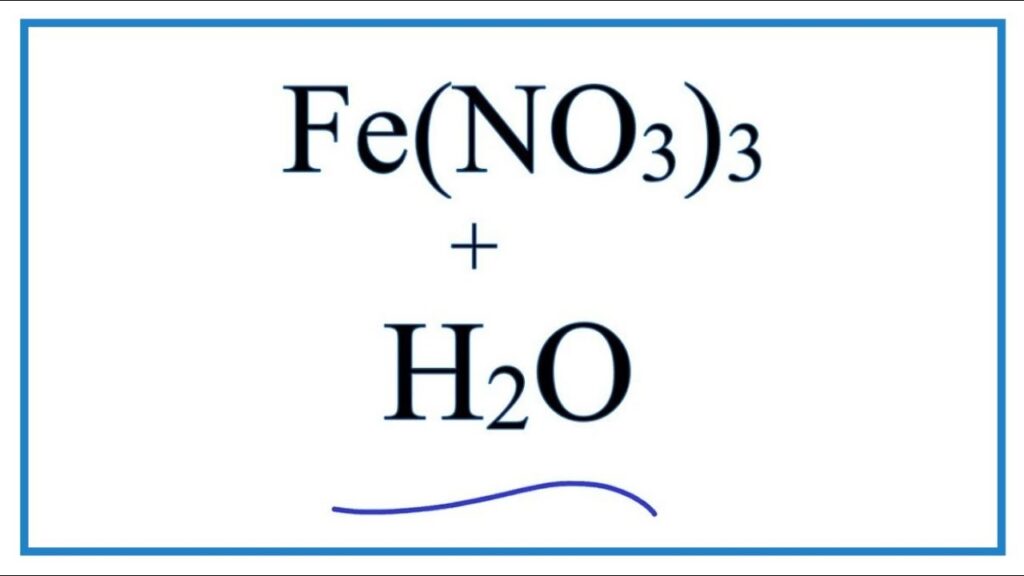
Ferric nitrate, also known as iron(III) nitrate, is a highly soluble inorganic chemical compound used in laboratories, industries, and water treatment. Its chemical formula is Fe(NO₃)₃, which represents iron in its +3 oxidation state combined with nitrate ions. This compound holds both scientific and industrial significance, making it a vital chemical in multiple sectors.
Ferric nitrate (Fe(NO₃)₃) is an important inorganic compound. For buyers, see our Ferric Nitrate Product Page for bulk supply and specifications.

Ferric Nitrate Formula and Structure
The formula for ferric nitrate is Fe(NO₃)₃.
- Fe stands for iron, in the +3 oxidation state.
- NO₃⁻ represents nitrate ions, each carrying a -1 charge.
- Since iron has a +3 charge, it bonds with three nitrate ions, resulting in Fe(NO₃)₃.
Ferric nitrate commonly exists in a hydrated form, with nonahydrate (Fe(NO₃)₃·9H₂O) being the most widely available. This hydrated crystal appears as a violet to grayish-white solid that is highly soluble in water and alcohol.
Physical and Chemical Properties
- Molecular Formula: Fe(NO₃)₃
- Molar Mass: ~241.86 g/mol (anhydrous)
- Appearance: Crystalline, light violet to colorless solid (hydrated)
- Solubility: Easily dissolves in water, ethanol, and acetone
- Nature: Strong oxidizing agent
As an oxidizer, ferric nitrate can accelerate combustion when in contact with organic materials.
Common Applications of Ferric Nitrate
1. Laboratory Use
Ferric nitrate is widely used in analytical chemistry as a reagent. It helps identify thiocyanates and other compounds due to its ability to form intensely colored complexes.
2. Catalyst in Chemical Reactions
It acts as a catalyst in organic synthesis, particularly in nitration reactions, and as a precursor in producing iron oxide catalysts.
3. Water Treatment
In industrial water systems, ferric nitrate is used as a coagulant to remove suspended solids and impurities. Its solubility ensures effective treatment across varied conditions.
It serves as a coagulant in industrial water systems. Learn more about our Water Treatment Chemicals
4. Metal Finishing and Etching
Ferric nitrate solutions are applied in metal surface treatment, etching, and polishing, especially in the jewelry and electronics industries.
5. Other Uses
- Precursor in nanoparticle synthesis
- Oxidizer in specialty pyrotechnics
- Intermediate for manufacturing iron-based chemicals
a key precursor in synthesis alongside Ammonium Ferric Citrate and other iron salts
Safety and Handling
Ferric nitrate is classified as an oxidizing and corrosive substance.
- Hazards: May cause skin, eye, and respiratory irritation. Strong oxidizer—fire risk when combined with organic materials.
- Precautions: Always handle with protective gloves, goggles, and proper ventilation. Store in a cool, dry place, away from reducing agents and flammable substances.
- Documentation: Safety Data Sheets (MSDS) must accompany industrial shipments.
All shipments include COA & MSDS, similar to our Sodium Cyanide Product Page.
Conclusion
The ferric nitrate formula, Fe(NO₃)₃, reflects its structure as an iron(III) salt of nitric acid. Its versatility—from water treatment and catalysis to laboratory analysis—makes it a crucial compound in chemistry and industry. When handled safely, ferric nitrate continues to provide valuable solutions across scientific and industrial applications.
Ferric nitrate continues to serve science and industry. To source high-quality material, visit our Ferric Nitrate Supplier Page.



